what portion of the lipoprotein is able to interact with both water and lipid molecules?
ABSTRACT
Cholesterol and triglycerides are insoluble in h2o and therefore these lipids must exist transported in association with proteins. Lipoproteins are complex particles with a central core containing cholesterol esters and triglycerides surrounded by free cholesterol, phospholipids, and apolipoproteins, which facilitate lipoprotein germination and function. Plasma lipoproteins tin can be divided into seven classes based on size, lipid limerick, and apolipoproteins (chylomicrons, chylomicron remnants, VLDL, IDL, LDL, HDL, and Lp (a)). Chylomicron remnants, VLDL, IDL, LDL, and Lp (a) are all pro-atherogenic while HDL is anti-atherogenic. Apolipoproteins have four major functions including ane) serving a structural function, ii) acting as ligands for lipoprotein receptors, iii) guiding the formation of lipoproteins, and 4) serving as activators or inhibitors of enzymes involved in the metabolism of lipoproteins. The exogenous lipoprotein pathway starts with the incorporation of dietary lipids into chylomicrons in the intestine. In the apportionment, the triglycerides carried in chylomicrons are metabolized in musculus and adipose tissue by lipoprotein lipase releasing free fatty acids, which are afterwards metabolized by muscle and adipose tissue, and chylomicron remnants are formed. Chylomicron remnants are and then taken upward past the liver. The endogenous lipoprotein pathway begins in the liver with the germination of VLDL. The triglycerides carried in VLDL are metabolized in muscle and adipose tissue past lipoprotein lipase releasing gratis fat acids and IDL are formed. The IDL are further metabolized to LDL, which are taken upwardly by the LDL receptor in numerous tissues including the liver, the predominant site of uptake. Opposite cholesterol transport begins with the formation of nascent HDL by the liver and intestine. These small HDL particles can then acquire cholesterol and phospholipids that are effluxed from cells, a process mediated by ABCA1 resulting in the formation of mature HDL. Mature HDL can acquire addition cholesterol from cells via ABCG1, SR-B1, or passive diffusion. The HDL then transports the cholesterol to the liver either directly by interacting with hepatic SR-B1 or indirectly by transferring the cholesterol to VLDL or LDL, a process facilitated by CETP. Cholesterol efflux from macrophages to HDL plays an of import role in protecting from the evolution of atherosclerosis. For complete coverage of all related areas of Endocrinology, please visit our on-line FREE web-text, Www.ENDOTEXT.ORG.
INTRODUCTION
Because lipids, such every bit cholesterol and triglycerides, are insoluble in water these lipids must exist transported in clan with proteins (lipoproteins) in the apportionment. Large quantities of fatty acids from meals must be transported as triglycerides to avoid toxicity. These lipoproteins play a key role in the assimilation and send of dietary lipids past the small intestine, in the transport of lipids from the liver to peripheral tissues, and the transport of lipids from peripheral tissues to the liver and intestine (reverse cholesterol send). A secondary function is to send toxic foreign hydrophobic and amphipathic compounds, such every bit bacterial endotoxin, from areas of invasion and infection (1).
Construction OF LIPOPROTEINS (two)
Lipoproteins are complex particles that have a fundamental hydrophobic core of not-polar lipids, primarily cholesterol esters and triglycerides. This hydrophobic core is surrounded by a hydrophilic membrane consisting of phospholipids, gratis cholesterol, and apolipoproteins (Figure 1). Plasma lipoproteins are divided into seven classes based on size, lipid composition, and apolipoproteins (Table i and Effigy two).

Effigy 1.
Lipoprotein Structure (figure modified from Biochemistry 39: 9763, 2000)
Table 1.
Lipoprotein Classes
View in ain window
| Lipoprotein | Density (g/ml) | Size (nm) | Major Lipids | Major Apoproteins |
|---|---|---|---|---|
| Chylomicrons | <0.930 | 75-1200 | Triglycerides | Apo B-48, Apo C, Apo Eastward, Apo A-I, A-2, A-Iv |
| Chylomicron Remnants | 0.930- i.006 | thirty-80 | Triglycerides Cholesterol | Apo B-48, Apo E |
| VLDL | 0.930- 1.006 | 30-80 | Triglycerides | Apo B-100, Apo E, Apo C |
| IDL | 1.006- 1.019 | 25-35 | Triglycerides Cholesterol | Apo B-100, Apo E, Apo C |
| LDL | i.019- 1.063 | 18- 25 | Cholesterol | Apo B-100 |
| HDL | i.063- i.210 | five- 12 | Cholesterol Phospholipids | Apo A-I, Apo A-Two, Apo C, Apo Eastward |
| Lp (a) | one.055- 1.085 | ~30 | Cholesterol | Apo B-100, Apo (a) |

Effigy 2:
Classes of Lipoproteins (effigy modified from Advances Protein Chemistry 45:303, 1994)
Chylomicrons
These are big triglyceride rich particles made by the intestine, which are involved in the transport of dietary triglycerides and cholesterol to peripheral tissues and liver. These particles contain apolipoproteins A-I, A-II, A-4, A-V, B-48, C-Two, C-III, and Eastward. Apo B-48 is the core structural poly peptide and each chylomicron particle contains one Apo B-48 molecule. The size of chylomicrons varies depending on the amount of fat ingested. A high fatty repast leads to the formation of big chylomicron particles due to the increased corporeality of triglyceride being transported whereas in the fasting land the chylomicron particles are small-scale carrying decreased quantities of triglyceride.
Chylomicron Remnants
The removal of triglyceride from chylomicrons by peripheral tissues results in smaller particles called chylomicron remnants. Compared to chylomicrons these particles are enriched in cholesterol and are pro-atherogenic.
Very Low-Density Lipoproteins (VLDL)
These particles are produced by the liver and are triglyceride rich. They comprise apolipoprotein B-100, C-I, C-II, C-III, and E. Apo B-100 is the core structural protein and each VLDL particle contains ane Apo B-100 molecule. Similar to chylomicrons the size of the VLDL particles can vary depending on the quantity of triglyceride carried in the particle. When triglyceride production in the liver is increased, the secreted VLDL particles are large. However, VLDL particles are smaller than chylomicrons.
Depression-Density Lipoproteins (LDL)
These particles are derived from VLDL and IDL particles and they are fifty-fifty further enriched in cholesterol. LDL carries the majority of the cholesterol that is in the circulation. The predominant apolipoprotein is B-100 and each LDL particle contains ane Apo B-100 molecule. LDL consists of a spectrum of particles varying in size and density. An abundance of pocket-sized dumbo LDL particles are seen in association with hypertriglyceridemia, depression HDL levels, obesity, type 2 diabetes (i.e. patients with the metabolic syndrome) and infectious and inflammatory states. These small dense LDL particles are considered to exist more pro-atherogenic than big LDL particles for a number of reasons. Minor dense LDL particles have a decreased affinity for the LDL receptor resulting in a prolonged retentiveness time in the apportionment. Additionally, they more easily enter the arterial wall and bind more than avidly to intra-arterial proteoglycans, which traps them in the arterial wall. Finally, pocket-size dense LDL particles are more susceptible to oxidation, which could result in an enhanced uptake by macrophages.
High-Density Lipoproteins (HDL)
These particles play an important role in reverse cholesterol transport from peripheral tissues to the liver, which is one potential machinery by which HDL may be anti-atherogenic. In addition, HDL particles take anti-oxidant, anti-inflammatory, anti-thrombotic, and anti-apoptotic properties, which may also contribute to their power to inhibit atherosclerosis. HDL particles are enriched in cholesterol and phospholipids. Apolipoproteins A-I, A-Two, A-IV, C-I, C-II, C-Three, and E are associated with these particles. Apo A-I is the core structural protein and each HDL particle may contain multiple Apo A-I molecules. HDL particles are very heterogeneous and tin can be classified based on density, size, charge, or apolipoprotein limerick (Table ii).
Table 2.
Nomenclature of HDL
View in ain window
| Method of nomenclature | Types of HDL |
|---|---|
| Density gradient ultracentrifugation | HDL2, HDL3, Very high-density HDL |
| Nuclear magnetic resonance | big, medium, and small-scale |
| Slope gel electrophoresis | HDL 2a, 2b, 3a, 3b, 3c |
| 2-dimensional gel electrophoresis | pre-beta ane and ii, alpha 1, 2, 3, iv |
| Apolipoprotein composition | A-I particles, A-I: A-Ii particles, A-I: E particles |
Lipoprotein (a) (Lp (a))
Lp (a) is an LDL particle that has apolipoprotein (a) attached to Apo B-100 via a disulfide bail. This particle is pro-atherogenic. The physiologic function of this lipoprotein is unknown.
APOLIPOPROTEINS (3,4)
Apolipoproteins have four major functions including one) serving a structural role, 2) acting as ligands for lipoprotein receptors, iii) guiding the formation of lipoproteins, and iv) serving equally activators or inhibitors of enzymes involved in the metabolism of lipoproteins (Tabular array 3). Apolipoproteins thus play a crucial office in lipoprotein metabolism.
Apolipoprotein A-I
Apo A-I is synthesized in the liver and intestine and is the major structural protein of HDL bookkeeping for approximately 70% of HDL protein. Information technology also plays a function in the interaction of HDL with ATP-bounden cassette protein A1 (ABCA1), ABCG1, and class B, type I scavenger receptor (SR-B1). Apo A-I is an activator of lecithin: cholesterol acyltransferase (LCAT), an enzyme that converts free cholesterol into cholesteryl ester. High levels of Apo A-I is associated with a decreased adventure of atherosclerosis.
Apolipoprotein A-Ii
Apo A-Two is synthesized in the liver and is the second about arable protein on HDL accounting for approximately 20% of HDL protein. The role of Apo A-2 in lipid metabolism is unclear. Apo A-2 is a strong predictor of risk for CVD.
Apolipoprotein A-IV (5)
Apo A-Four is synthesized in the intestine during fat absorption. Apo A-IV is associated with chylomicrons and high-density lipoproteins, but is also found in the lipoprotein-free fraction. Its precise role in lipoprotein metabolism remains to be determined but studies have suggested a function for Apo A-4 in regulating food intake.
Apolipoprotein A-V (6)
Apo A-V is synthesized in the liver and associates with triglyceride rich lipoproteins. Information technology is an activator of LPL mediated lipolysis and thereby plays an important office in the metabolism of triglyceride rich lipoproteins.
Apolipoprotein B-48
Apo B-48 is synthesized in the intestine and is the major structural protein of chylomicrons and chylomicron remnants. There is a single molecule of apo B-48 per chylomicron particle. There is a single apolipoprotein B gene that is expressed in both the liver and intestine. The intestine expresses a protein that is approximately ½ the size of the liver due to mRNA editing. The apobec-ane editing complex is expressed in the intestine and edits a specific cytidine to an uracil in the apo B mRNA in the intestine creating a terminate codon that results in the cessation of poly peptide translation and a shorter Apo B (Apo B-48). Notably Apo B-48 is not recognized by the LDL receptor.
Apolipoprotein B-100
Apo B-100 is synthesized in the liver and is the major structural component of VLDL, IDL, and LDL. At that place is a single molecule of Apo B-100 per VLDL, IDL, and LDL particle. Apo B-100 is a ligand for the LDL receptor and therefore plays an important role in the clearance of lipoprotein particles. Loftier levels of Apo B-100 is associated with an increased risk of atherosclerosis.
Apolipoprotein C (7,viii)
The C apolipoproteins are synthesized primarily in the liver and freely substitution betwixt lipoprotein particles and therefore are found in clan with chylomicrons, VLDL, and HDL.
Apo C-2 is a co-factor for lipoprotein lipase (LPL) and thus stimulates triglyceride hydrolysis (7). Loss of role mutations in Apo C-Two effect in marked hypertriglyceridemia due to a failure to metabolize triglyceride rich lipoproteins.
Apo C-Iii is an inhibitor of LPL (nine). Additionally, Apo C-3 inhibits the interaction of triglyceride rich lipoproteins with their receptors. Recent studies take shown that loss of office mutations in Apo C-Three pb to decreases in serum triglyceride levels and a reduced risk of cardiovascular disease. Interestingly, inhibition of Apo C-III expression results in a subtract in serum triglyceride levels fifty-fifty in patients deficient in lipoprotein lipase indicating that the power of Apo C-Iii to modulate serum triglyceride levels is not dependent solely on regulating lipoprotein lipase activity.
Apolipoprotein Eastward (10)
Apolipoprotein Eastward is synthesized in many tissues but the liver and intestine are the primary source of circulating Apo Eastward. Apo E exchanges betwixt lipoprotein particles and is associated with chylomicrons, chylomicron remnants, VLDL, IDL, and a subgroup of HDL particles. In that location are three common genetic variants of Apo E (Apo E2, E3, and E4). ApoE2 differs from the most common isoform, Apo E3, past a unmarried amino acid substitution where cysteine substitutes for arginine at residue 158. Apo E4 differs from Apo E3 at residue 112, where arginine substitutes for cysteine. Apo E3 and E4 are ligands for the LDL receptor while Apo E2 is poorly recognized by the LDL receptor. Patients who are homozygous for Apo E2 can develop familial dysbetalipoproteinemia. Apo E4 is associated with an increased gamble of Alzheimer'due south disease and an increased gamble of atherosclerosis.
Apolipoprotein (a) (11)
Apo (a) is synthesized in the liver. This protein is a homolog of plasminogen and its molecular weight varies from 300,000 to 800,000. It is fastened to Apo B-100 via a disulfide bail. Loftier levels of Apo (a) are associated with an increased take a chance of atherosclerosis. Apo (a) is an inhibitor of fibrinolysis and can also heighten the uptake of lipoproteins by macrophages, both of which could increase the risk of atherosclerosis. The physiologic function of Apo (a) is unknown. Interestingly this apolipoprotein is found in primates but not in other species.
Table 3.
Apolipoproteins
View in own window
| Apolipoprotein | MW | Primary Source | Lipoprotein Clan | Function |
|---|---|---|---|---|
| Apo A-I | 28,000 | Liver, Intestine | HDL, chylomicrons | Structural protein for HDL, Activates LCAT |
| Apo A-Two | 17,000 | Liver | HDL, chylomicrons | Structural protein for HDL, Activates hepatic lipase |
| Apo A-Four | 45,000 | Intestine | HDL, chylomicrons | Unknown |
| Apo A-5 | 39,000 | Liver | VLDL, chylomicrons, HDL | Promotes LPL mediated TG lipolysis |
| Apo B-48 | 241,000 | Intestine | Chylomicrons | Structural poly peptide for chylomicrons |
| Apo B-100 | 512,000 | Liver | VLDL, IDL, LDL, Lp (a) | Structural poly peptide, Ligand for LDL receptor |
| Apo C-I | 6,600 | Liver | Chylomicrons, VLDL, HDL | Activates LCAT |
| Apo C-Two | 8,800 | Liver | Chylomicrons, VLDL, HDL | Co-factor for LPL |
| Apo C-Iii | 8,800 | Liver | Chylomicrons, VLDL, HDL | Inhibits LPL and uptake of lipoproteins |
| Apo E | 34,000 | Liver | Chylomicron remnants, IDL, HDL | Ligand for LDL receptor |
| Apo (a) | 250,000- 800,00 | Liver | Lp (a) | Inhibits plasminogen activation |
LIPOPROTEIN RECEPTORS AND LIPID TRANSPORTERS
There are several receptors and transporters that play a crucial part in lipoprotein metabolism.
LDL Receptor (12)
The LDL receptor is present in the liver and virtually other tissues. It recognizes Apo B-100 and Apo E and hence mediates the uptake of LDL, chylomicron remnants, and IDL, which occurs via endocytosis (Figure 3). After internalization, the lipoprotein particle is degraded in lysosomes and the cholesterol is released. The delivery of cholesterol to the jail cell decreases the activity of HMGCoA reductase, a fundamental enzyme in the biosynthesis of cholesterol, and the expression of LDL receptors. LDL receptors in the liver play a major role in determining plasma LDL levels (a low number of receptors is associated with high plasma LDL levels while a high number of hepatic LDL receptors is associated with depression plasma LDL levels). The number of LDL receptors is regulated past the cholesterol content of the cell (13). When cellular cholesterol levels are decreased the transcription gene SREBP is transported from the endoplasmic reticulum to the golgi where proteases cleave and activate SREBP, which then migrates to the nucleus and stimulates the expression of LDL receptors (Figure 4). Conversely, when cellular cholesterol levels are high SREBP remains in the endoplasmic reticulum in an inactive form and the expression of LDL receptors is low. As discussed later PCSK9 regulates the rate of deposition of LDL receptors.
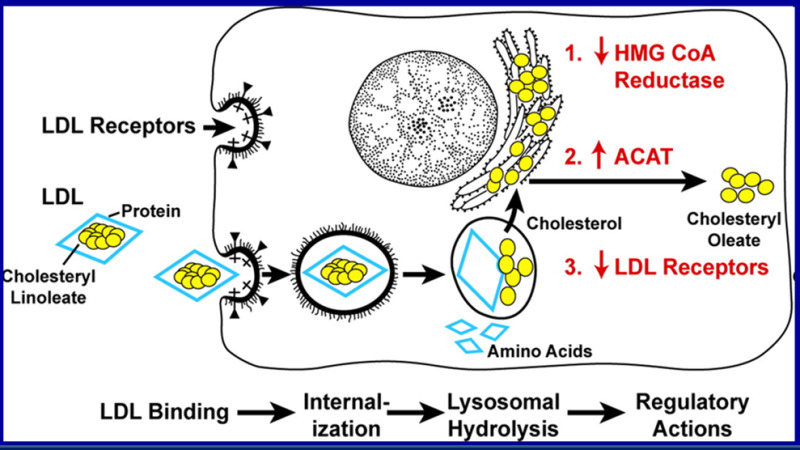
Figure 3.
LDL Receptor Pathway (figure modified from Annual Review of Biochemistry 46: 897, 1977)

Figure 4.
SREBP Pathway (figure modified from Periodical of Lipid Inquiry fifty: Supp S15, 2009)
Form B Scavenger Receptor B1 (SR-B1) (15)
SR-B1 is expressed in the liver, adrenal glands, ovaries, testes, macrophages, and other cells. In the liver and steroid producing cells, it mediates the selective uptake of cholesterol esters from HDL particles. In macrophages and other cells, it facilitates the efflux of cholesterol from the jail cell to HDL particles.
ATP-Binding Cassette Transporter A1 (ABCA1) (16)
ABCA1 is expressed in many cells including hepatocytes, enterocytes, and macrophages. It mediates the transport of cholesterol and phospholipids from the prison cell to lipid poor HDL particles (pre-beta-HDL).
ATP-Binding Cassette Transporter G1 (ABCG1) (17)
ABCG1 is expressed in many different cell types and mediates the efflux of cholesterol from the cell to HDL particles.
ATP-Binding Cassette Transporter G5 and G8 (ABCG5/ABCG8) (xviii)
ABCG5 and ABCG8 are expressed in the liver and intestine and grade a heterodimer. In the intestine, these transporters mediate the motion of plant sterols and cholesterol from inside the enterocyte into the intestinal lumen thereby decreasing their absorption and limiting the uptake of dietary plant sterols. In the liver, these transporters play a role in the motility of cholesterol and plant sterols into the bile facilitating the excretion of found sterols.
Niemann-Selection C1-Similar 1 (NPC1L1) (18)
NPC1L1 is expressed in the intestine and mediates the uptake of cholesterol and plant sterols from the abdominal lumen into the enterocyte.
ENZYMES AND TRANSFER PROTEINS INVOLVED IN LIPOPROTEIN METABOLISM
At that place are several enzymes and transfer proteins that play a central role in lipoprotein metabolism.
Lipoprotein Lipase (LPL) (xix)
LPL is synthesized in muscle, heart, and adipose tissue, then secreted and attached to the endothelium of the next blood capillaries. This enzyme hydrolyzes the triglycerides carried in chylomicrons and VLDL to fatty acids, which can be taken up by cells. The catabolism of triglycerides results in the conversion of chylomicrons into chylomicron remnants and VLDL into IDL. This enzyme requires Apo C-Two as a cofactor. Apo A-5 also plays a cardinal office in the activation of this enzyme. In contrast Apo C-III and Apo A-II inhibit the activity of LPL. Insulin stimulates LPL expression and LPL activeness is reduced in patients with poorly controlled diabetes, which can impair the metabolism of triglyceride rich lipoproteins leading to hypertriglyceridemia.
Hepatic Lipase (twenty)
Hepatic lipase is localized to the sinusoidal surface of liver cells. It mediates the hydrolysis of triglycerides and phospholipids in IDL and LDL leading to smaller particles (IDL is converted to LDL; LDL is converted from large LDL to small LDL). It also mediates the hydrolysis of triglycerides and phospholipids in HDL resulting in smaller HDL particles.
Endothelial Lipase (21)
This lipase plays a major function in hydrolyzing the phospholipids in HDL.
Lecithin: Cholesterol Acyltransferase (LCAT) (22)
LCAT is made in the liver. In the plasma, it catalyzes the synthesis of cholesterol esters in HDL past facilitating the transfer of a fatty acid from position two of lecithin to cholesterol. This allows for the transfer of the cholesterol from the surface of the HDL particle (gratis cholesterol) to the core of the HDL particle (cholesterol ester), which facilitates the continued uptake of free cholesterol by HDL particles by reducing the concentration of cholesterol on the surface of HDL.
Cholesteryl Ester Transfer Protein (CETP) (23)
This protein is synthesized in the liver and in the plasma mediates the transfer of cholesterol esters from HDL to VLDL, chylomicrons, and LDL and the transfer of triglycerides from VLDL and chylomicrons to HDL. Inhibition of CETP activity leads to an increase in HDL cholesterol and a decrease in LDL cholesterol.
EXOGENOUS LIPOPROTEIN PATHWAY (CHYLOMICRONS)

Figure 5.
Exogenous Lipoprotein Pathway
Fat Assimilation (24-27)
The exogenous lipoprotein pathway starts in the intestine. Dietary triglycerides (approximately 100 grams per solar day) are hydrolyzed to free fat acids and monoacylglycerol past intestinal lipases and emulsified with bile acids, cholesterol, plant sterols, and fat-soluble vitamins to form micelles. While the fatty acids in the intestine are overwhelmingly accounted for by dietary intake the cholesterol in the intestinal lumen is primarily derived from bile (approximately 800-1200mg of cholesterol from bile vs. 300-500mg from diet). Plant sterols account for approximately 25% of dietary sterol intake (approximately 100-150mg/day). The cholesterol, found sterols, fat acids, monoacylglycerol, and fatty-soluble vitamins contained in the micelles are so transported into the intestinal cells. The uptake of cholesterol and plant sterols from the intestinal lumen into intestinal cells is facilitated by a sterol transporter, Niemann-Pick C1- like 1 protein (NPC1L1) (Figure 6). Ezetimibe, a drug which inhibits intestinal cholesterol and plant sterol uptake, binds to NPC1L1 and inhibits its activeness. In one case in the abdominal cell the cholesterol and plant sterols may be transported back into the intestinal lumen, a process mediated by ABCG5 and ABCG8, or converted to sterol esters by acyl-CoA cholesterol acyl transferase (ACAT), which attaches a fatty acid to the sterol. Compared to cholesterol, plant sterols are poor substrates for ACAT and therefore the germination of found sterol esters does not occur equally efficiently as the germination of cholesterol esters. In humans, <5% of dietary plant sterols are absorbed and the vast majority are transported out of the intestine cell, a process mediated by ABCG5 and ABCG8, which are very efficient at effluxing plant sterols from the intestinal cell into the intestinal lumen. Patients with sitosterolemia take mutations in either ABCG5 or ABCG8 and net absorption of dietary plant sterols is increased (20-30% absorbed vs. < v% in normal subjects). Thus, ABCG5 and ABCG8 along with ACAT serve equally gate keepers and cake the uptake of constitute sterols and probable also play an of import role in determining the efficiency of cholesterol absorption (humans typically blot only approximately l% of dietary cholesterol with a range of 25-75%).
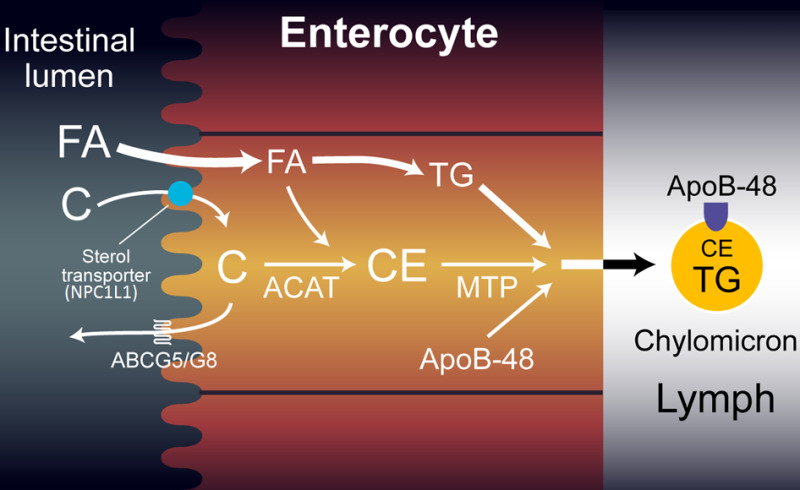
Figure 6.
Intestinal Cell and Sterol Metabolism
The pathway of absorption of costless fatty acids is non well understood but it is likely that both passive diffusion and specific transporters play a role. The fatty acid transporter CD36 is strongly expressed in the proximal 3rd of the intestine and is localized to the villi. While this transporter probable plays a part in fatty acid uptake past intestinal cells, this transporter is not essential equally humans and mice deficient in this poly peptide exercise non have fat malabsorption. However, in mice deficient in CD36 there is a shift in the absorption of lipid to the distal intestine, suggesting pathways that can compensate for the absenteeism of CD36. Fatty acid transport protein 4 (FATP4) is too highly expressed in the intestine. Nonetheless, mice deficient in FATP4 exercise non have abnormalities in fat absorption. The pathways by which monoacylglycerols are absorbed past intestinal cells remain to be defined.
Formation of Chylomicrons (24,27)
The absorbed fatty acids and monoacylglycerols are utilized to synthesize triglycerides. The key enzymes required for triglyceride synthesis are monoacylglycerol acyltransferase (MGAT) and diacylglycerol transferase (DGAT). MGAT catalyzes the addition of a fatty acid to monoacylglycerol while DGAT catalyzes the improver of a fatty acid to diacylglycerol resulting in triglyceride germination. As noted in a higher place, the majority of the cholesterol absorbed by the intestine is esterified to cholesterol esters by ACAT. The triglycerides and cholesterol esters are packaged into chylomicrons in the endoplasmic reticulum. The size and composition of the chylomicrons formed in the intestine are dependent on the amount of fat ingested and absorbed by the intestine and the type of fatty absorbed. Increased fat assimilation results in larger chylomicrons. The germination of chylomicrons in the endoplasmic reticulum requires the synthesis of Apo B-48 by the intestinal cell (Figure half dozen). Microsomal triglyceride transfer protein (MTP) is required for the movement of lipid from the endoplasmic reticulum to the Apo B-48. The absence of MTP results in the disability to form chylomicrons (Abetalipoproteinemia). Lomitapide inhibits MTP role and is used to treat patients with homozygous Familial Hypercholesterolemia.
ENDOGENOUS LIPOPROTEIN PATHWAY (VLDL AND LDL)
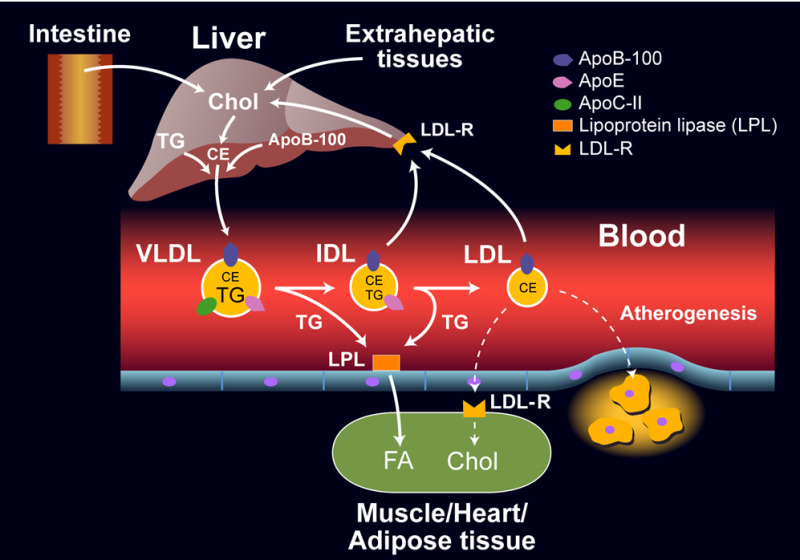
Figure vii.
Endogenous Lipoprotein Pathway
Formation of VLDL (32,33)
In the liver triglycerides and cholesterol esters are transferred in the endoplasmic reticulum to newly synthesized Apo B-100. Similar to the intestine this transfer is mediated by MTP. The availability of triglycerides is the primary determinant of the rate of VLDL synthesis. If the supply of triglyceride is limited the newly synthesized Apo B is rapidly degraded. Thus, in contrast to many proteins the rate of synthesis of the Apo B-100 is not the major determinant of the charge per unit of secretion. Rather the corporeality of lipid available determines whether Apo B-100 is degraded or secreted. MTP is required for the early improver of lipid to Apo B-100 particles but additional lipid is added via pathways that do non crave MTP. Loss of function mutations in either Apo B-100 or MTP consequence in the failure to produce VLDL and marked decreases in plasma triglyceride and cholesterol levels (Familial hypobetalipoproteinemia or abetalipoproteinemia). The precise pathway by which the newly synthesized VLDL particles are secreted from the hepatocyte into the circulation is not resolved.
LIPOPROTEIN (a) (xi,45,46)
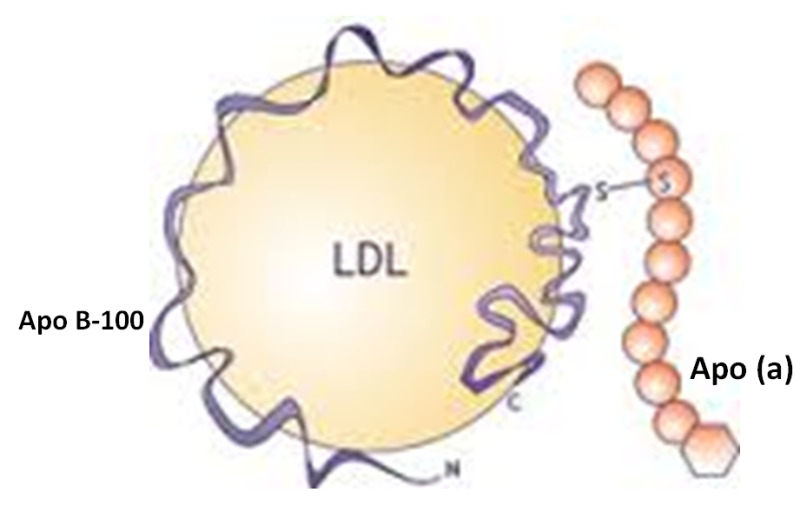
Figure 10.
Lp (a)
Lp (a) consists of an LDL molecule and a unique apolipoprotein (a), which is fastened to the Apo B-100 of the LDL via a single disulfide bound. Lp (a) contain Apo (a) and Apo B-100 in a 1:ane tooth ratio. Like Apo B-100, apo (a) is also made by hepatocytes. Apo (a) contains multiple kringle motifs that are like to the kringle repeats in plasminogen. The number of kringle repeats tin can vary and thus the molecular weight of apo (a) can range from 250,000 to 800,000. The levels of Lp (a) in plasma tin vary more a 1000-fold ranging from undetectable to greater than 100mg/dl. Lp (a) levels largely reflect Lp (a) production rates, which are primarily genetically regulated. Individuals with high molecular weight Apo (a) proteins tend to have lower levels of Lp (a) while individuals with low molecular weight Apo (a) tend to take college levels. It is hypothesized that the liver is less efficient in secreting high molecular weight Apo (a). The mechanism of Lp (a) clearance is uncertain merely does non appear to involve LDL receptors. Elevated plasma Lp(a) levels are associated with an increased run a risk of atherosclerosis. Therapies that advance LDL clearance and lower LDL levels do not lower Lp (a) levels (for instance statin therapy). The kidney appears to play an important role in Lp (a) clearance as kidney disease is associated with delayed clearance and elevations in Lp (a) levels.
REFERENCES
- 1.
-
Feingold KR, Grunfeld C. Lipids: a key player in the battle betwixt the host and microorganisms. J Lipid Res. 2012;53:2487–2489. [PMC free article: PMC3494250] [PubMed: 23075464]
- 2.
-
Smith LC, Pownall HJ, Gotto AM Jr. The plasma lipoproteins: structure and metabolism. Annu Rev Biochem. 1978;47:751–757. [PubMed: 209732]
- 3.
-
Mahley RW, Innerarity TL, Rall SC Jr, Weisgraber KH. Plasma lipoproteins: apolipoprotein construction and function. J Lipid Res. 1984;25:1277–1294. [PubMed: 6099394]
- 4.
-
Breslow JL. Human apolipoprotein molecular biological science and genetic variation. Annu Rev Biochem. 1985;54:699–727. [PubMed: 3896129]
- 5.
-
Wang F, Kohan AB, Lo CM, Liu One thousand, Howles P, Tso P. Apolipoprotein A-IV: a protein intimately involved in metabolism. J Lipid Res. 2015;56:1403–1418. [PMC free commodity: PMC4513983] [PubMed: 25640749]
- 6.
- 7.
-
Wolska A, Dunbar RL, Freeman LA, Ueda Chiliad, Amar MJ, Sviridov Practice, Remaley AT. Apolipoprotein C-Ii: New findings related to genetics, biochemistry, and role in triglyceride metabolism. Atherosclerosis. 2017;267:49–60. [PMC gratis article: PMC5705268] [PubMed: 29100061]
- 8.
-
Ramms B, Gordts P. Apolipoprotein C-III in triglyceride-rich lipoprotein metabolism. Curr Opin Lipidol. 2018;29:171–179. [PubMed: 29547399]
- 9.
-
Taskinen MR, Boren J. Why Is Apolipoprotein CIII Emerging as a Novel Therapeutic Target to Reduce the Burden of Cardiovascular Disease? Curr Atheroscler Rep. 2016;18:59. [PMC free article: PMC5018018] [PubMed: 27613744]
- x.
- eleven.
-
Nordestgaard BG, Langsted A. Lipoprotein (a) as a cause of cardiovascular disease: insights from epidemiology, genetics, and biology. J Lipid Res. 2016;57:1953–1975. [PMC gratuitous article: PMC5087876] [PubMed: 27677946]
- 12.
- 13.
-
Goldstein JL, DeBose-Boyd RA, Chocolate-brown MS. Protein sensors for membrane sterols. Cell. 2006;124:35–46. [PubMed: 16413480]
- 14.
-
van de Sluis B, Wijers M, Herz J. News on the molecular regulation and office of hepatic depression-density lipoprotein receptor and LDLR-related protein 1. Curr Opin Lipidol. 2017;28:241–247. [PMC costless article: PMC5482905] [PubMed: 28301372]
- 15.
-
Trigatti BL. SR. -B1 and PDZK1: partners in HDL regulation. Curr Opin Lipidol. 2017;28:201–208. [PubMed: 28134663]
- sixteen.
- 17.
-
Baldan A, Tarr P, Lee R, Edwards PA. ATP-binding cassette transporter G1 and lipid homeostasis. Curr Opin Lipidol. 2006;17:227–232. [PubMed: 16680026]
- 18.
-
Kidambi S, Patel SB. Cholesterol and non-cholesterol sterol transporters: ABCG5, ABCG8 and NPC1L1: a review. Xenobiotica. 2008;38:1119–1139. [PubMed: 18668442]
- nineteen.
-
Olivecrona 1000. Role of lipoprotein lipase in lipid metabolism. Curr Opin Lipidol. 2016;27:233–241. [PubMed: 27031275]
- twenty.
-
Kobayashi J, Miyashita K, Nakajima Yard, Mabuchi H. Hepatic Lipase: a Comprehensive View of its Part on Plasma Lipid and Lipoprotein Metabolism. J Atheroscler Thromb. 2015;22:1001–1011. [PubMed: 26194979]
- 21.
-
Yasuda T, Ishida T, Rader DJ. Update on the role of endothelial lipase in loftier-density lipoprotein metabolism, opposite cholesterol transport, and atherosclerosis. Circ J. 2010;74:2263–2270. [PubMed: 20962428]
- 22.
-
Ossoli A, Simonelli S, Vitali C, Franceschini Yard, Calabresi L. Function of LCAT in Atherosclerosis. J Atheroscler Thromb. 2016;23:119–127. [PubMed: 26607351]
- 23.
-
Mabuchi H, Nohara A, Inazu A. Cholesteryl ester transfer protein (CETP) deficiency and CETP inhibitors. Mol Cells. 2014;37:777–784. [PMC complimentary article: PMC4255097] [PubMed: 25410905]
- 24.
- 25.
-
D'Aquila T, Hung YH, Carreiro A, Buhman KK. Contempo discoveries on absorption of dietary fat: Presence, synthesis, and metabolism of cytoplasmic lipid droplets inside enterocytes. Biochim Biophys Acta. 2016;1861:730–747. [PMC free commodity: PMC5503208] [PubMed: 27108063]
- 26.
- 27.
-
Kindel T, Lee DM, Tso P. The mechanism of the formation and secretion of chylomicrons. Atheroscler Suppl. 2010;11:11–16. [PubMed: 20493784]
- 28.
-
Dallinga-Thie GM, Franssen R, Mooij HL, Visser ME, Hassing HC, Peelman F, Kastelein JJ, Peterfy Grand, Nieuwdorp M. The metabolism of triglyceride-rich lipoproteins revisited: new players, new insight. Atherosclerosis. 2010;211:1–8. [PMC free article: PMC3924774] [PubMed: 20117784]
- 29.
-
Dijk W, Kersten S. Regulation of lipid metabolism past angiopoietin-like proteins. Curr Opin Lipidol. 2016;27:249–256. [PubMed: 27023631]
- 30.
-
Fong LG, Immature SG, Beigneux AP, Bensadoun A, Oberer G, Jiang H, Ploug M. GPIHBP1 and Plasma Triglyceride Metabolism. Trends Endocrinol Metab. 2016;27:455–469. [PMC free article: PMC4927088] [PubMed: 27185325]
- 31.
-
Peterfy M. Lipase maturation cistron 1: a lipase chaperone involved in lipid metabolism. Biochim Biophys Acta. 2012;1821:790–794. [PMC free article: PMC3288453] [PubMed: 22063272]
- 32.
- 33.
-
Hooper AJ, Burnett JR, Watts GF. Gimmicky aspects of the biology and therapeutic regulation of the microsomal triglyceride transfer protein. Circ Res. 2015;116:193–205. [PubMed: 25552696]
- 34.
- 35.
- 36.
-
Zhang L, Reue K, Fong LG, Immature SG, Tontonoz P. Feedback regulation of cholesterol uptake by the LXR-IDOL-LDLR axis. Arterioscler Thromb Vasc Biol. 2012;32:2541–2546. [PMC free article: PMC4280256] [PubMed: 22936343]
- 37.
-
Brown MS, Radhakrishnan A, Goldstein JL. Retrospective on Cholesterol Homeostasis: The Central Part of Scap. Annual review of biochemistry. 2017 [PMC free commodity: PMC5828883] [PubMed: 28841344]
- 38.
-
Rosenson RS, Brewer HB Jr, Davidson WS, Fayad ZA, Fuster V, Goldstein J, Hellerstein M, Jiang Ninety, Phillips MC, Rader DJ, Remaley AT, Rothblat GH, Tall AR, Yvan-Charvet Fifty. Cholesterol efflux and atheroprotection: advancing the concept of reverse cholesterol transport. Circulation. 2012;125:1905–1919. [PMC complimentary article: PMC4159082] [PubMed: 22508840]
- 39.
- 40.
-
Zhao Y, Van Berkel TJ, Van Eck Grand. Relative roles of various efflux pathways in net cholesterol efflux from macrophage foam cells in atherosclerotic lesions. Curr Opin Lipidol. 2010;21:441–453. [PubMed: 20683325]
- 41.
-
Lee-Rueckert M, Escola-Gil JC, Kovanen PT. HDL functionality in reverse cholesterol transport--Challenges in translating data emerging from mouse models to human disease. Biochim Biophys Acta. 2016;1861:566–583. [PubMed: 26968096]
- 42.
-
Tall AR. Cholesterol efflux pathways and other potential mechanisms involved in the athero-protective result of high density lipoproteins. J Intern Med. 2008;263:256–273. [PubMed: 18271871]
- 43.
-
Siddiqi HK, Kiss D, Rader D. HDL-cholesterol and cardiovascular affliction: rethinking our approach. Curr Opin Cardiol. 2015;30:536–542. [PubMed: 26192490]
- 44.
-
Moore KJ, Rayner KJ, Suarez Y, Fernandez-Hernando C. The part of microRNAs in cholesterol efflux and hepatic lipid metabolism. Annu Rev Nutr. 2011;31:49–63. [PMC complimentary article: PMC3612434] [PubMed: 21548778]
- 45.
- 46.
-
Schmidt K, Noureen A, Kronenberg F, Utermann G. Structure, function, and genetics of lipoprotein (a). J Lipid Res. 2016;57:1339–1359. [PMC free article: PMC4959873] [PubMed: 27074913]
Source: https://www.ncbi.nlm.nih.gov/books/NBK305896/
0 Response to "what portion of the lipoprotein is able to interact with both water and lipid molecules?"
Publicar un comentario